Solution video
OpenStax College Physics, Chapter 1, Problem 20 (Problems & Exercises)

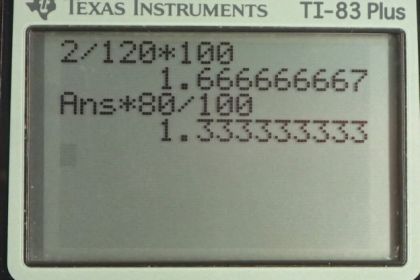
Calculator Screenshots
Comments
Why does the absolute uncertainty affect the sig figs. No matter what you will not get more sig figs since it's a whole number.
Hi Ren, I'll try to answer this, but I might need a better understanding of what you're asking. The absolute uncertainty of 2 mm has one significant figure, but the fact that it's a whole number isn't relevant. If the uncertainty had hypothetically been 0.2 mm, that would have one significant figure also. Sometimes errors are estimated with greater precision, as in another hypothetical example 2.0 mm, which has two significant figures. Had the absolute uncertainty been estimated as 2.0 mm, then the percent error calculated in part (a) would have been written as 1.7% instead of 2%, and part (b) would have been 1.3 mm instead of 1 mm.
If that doesn't answer your question, just let me know.
All the best,
Shaun