Solution video
OpenStax College Physics, Chapter 5, Problem 27 (Problems & Exercises)
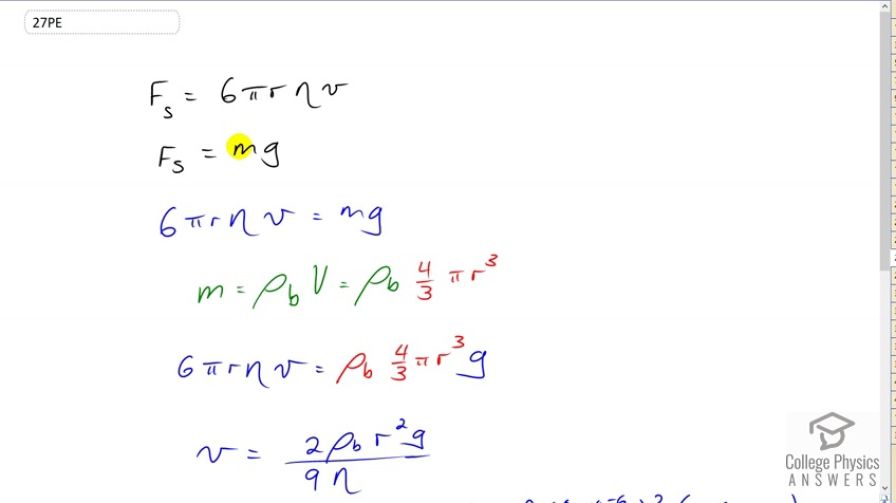
Calculator Screenshots
Comments
Where did the formula for mass come from? m=pbV=pb4/3pir^3?
I am also confused by this leap.
Hi georgeh, thanks for the question. Density is defined as mass per volume, which is written as . This can be rearranged to solve for mass by multiplying both sides by volume. Doing this makes . The in the video has a subscript b just as a label to say that it's the density of the bacterium. Then, since the bacterium is assumed to be a sphere, the volume is substituted with the formula for the volume of a sphere, which is . In the end this makes .
Hope this helps!
All the best,
Shaun
im confused as to where the '2pb' came from in the second last step for v
Are you at 1:15 in the video? I think the calculation you might be wondering about is where the comes from? That's the result of divided by .
Hope this helps,
Shaun
Thank you for your help on this question. I just needed a nudge and got the answer which was confirmed by yours. Any thoughts on what the threshold is for moving from the standard F(D) equation to the Stokes equation? The book just says "very small" and if you use the standard equation, you have an unknown for C in addition to v which stumped me for about 20 minutes...
Also quite distressing that the text assumes we know what viscosity is about when it isn't covered until chapter 12 and this question is in chapter 5 :)
Thanks for your time and assistance.



