Question
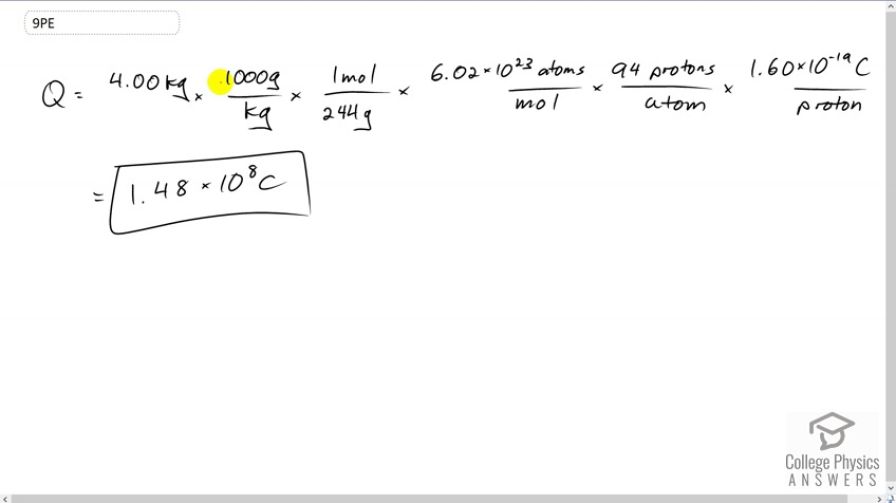
How many coulombs of positive charge are there in 4.00 kg of plutonium, given its atomic mass is 244 and that each plutonium atom has 94 protons?
Final Answer
Solution video
OpenStax College Physics, Chapter 18, Problem 9 (Problems & Exercises)
vote with a rating of
votes with an average rating of
.
Calculator Screenshots
Video Transcript
This is College Physics Answers with Shaun Dychko. We’re going to find the number of coulombs of positive charge that are in four kilograms of Plutonium. So the charge is going to be four kilograms times 1000 grams per kilogram, and we do this conversion because the molar mass has units of one mol for every 244 grams, when I say molar mass it’s the same as atomic mass but just written with units of mol here because I want to multiply by Avogadro's number next which is 6.02 times ten to the 23 atoms for every mol. And so far our unit conversion we’re at number of atoms here, and then we multiply by 94 protons for every atom, so now we have number of protons, and then we multiply by 1.6 times ten to the minus 19 coulombs per proton, now at long last we have units of coulombs, which is 1.48 times ten to the eight coulombs.

