Question
What is the atmospheric pressure on top of Mt. Everest on a day when water boils there at a temperature of ?
Final Answer
Solution video
OpenStax College Physics for AP® Courses, Chapter 13, Problem 53 (Problems & Exercises)
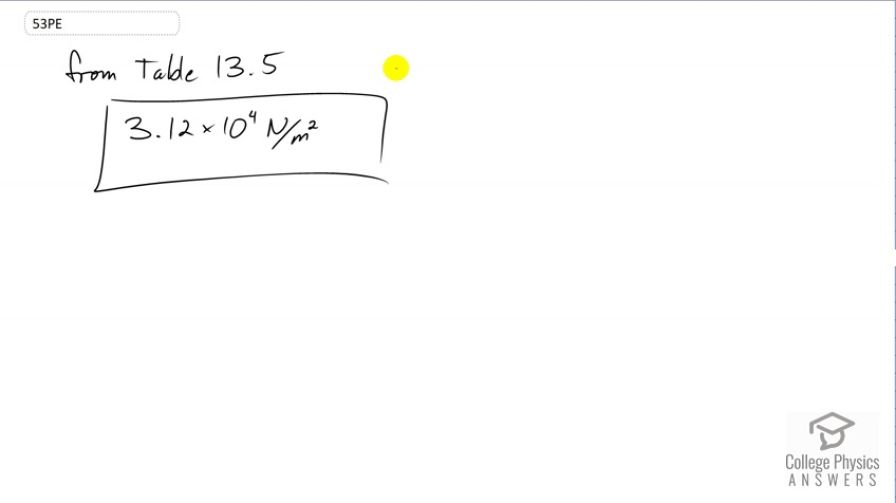
vote with a rating of
votes with an average rating of
.
Video Transcript
This is College Physics Answers with Shaun Dychko. Water will boil when its vapor pressure equals the surrounding atmospheric pressure because that surrounding atmospheric pressure is the maximum possible partial pressure of the water in the air. So, when we look at Table 13.5, we can see that at 70 degrees Celsius, the vapor pressure of water is 3.12 times ten to the four newtons per square meter and that will equal the atmospheric pressure. So that's what this number is.