Question
Write the complete decay equation for the given nuclide in the complete notation:
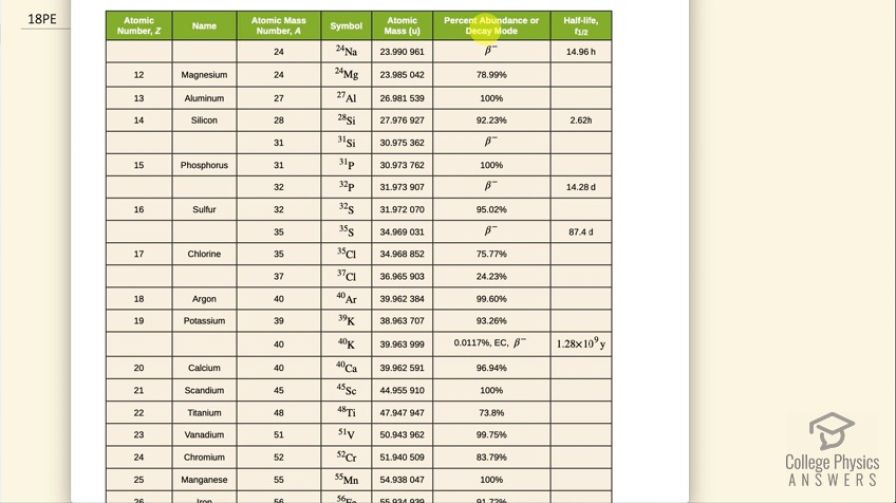
decay of , a naturally occurring rare isotope of potassium responsible for some of our exposure to
background radiation.
Final Answer
Please see the solution video.
Solution video
OpenStax College Physics for AP® Courses, Chapter 31, Problem 18 (Problems & Exercises)
vote with a rating of
votes with an average rating of
.
Video Transcript
This is College Physics Answers with Shaun Dychko. This is the formula for β decay of potassium-40. We look up in the appendix at the end of the text book what the atomic number is for potassium and it's 19 so there's 19 protons in potassium-40 and that means there are 21 neutrons and a neutron turns into a proton, an electron and an electron anti-neutrino during β decay. So that's why the number of neutrons is 1 less so we subtract 1 from the neutrons and we add 1 to the number of protons. So neutron turns into a proton and this electron which for historical reasons is called a β particle but it's the same as an electron zipping out as a result of this β decay and in order to have the electron family number conserved, there is an anti-electron neutrino also emitted.